Time-resolved evanescent wave-induced fluorescence spectroscopy
We use the evanescent field, generated upon the total internal reflection of light at the interface between two materials of differing refractive index, to induce fluorescence from fluorophores residing in close proximity to the interface. By using a mode-locked laser as the excitation source, and TCSPC for detection the emission is time resolved which can provide information on the dynamics and local environment of the fluorophore. These time-resolved EWIF measurements can be performed as a function the angle of incidence to probe changes in fluorescence decay behaviour as a function of distance from the interface.
Polarised EWIF Spectroscopy:
Another level of complexity in using the evanescent wave to induce emission is to use polarised excitation and emission measurements to extract information on emission depolarising processes (e.g. molecular rotation and energy migration) and differentiate between these processes occurring within and perpendicular to the plane of the interface. Excitation with s-polarised light will preferentially photoselect molecules with their absorption transition moments parallel to the plane of the interface, whereas p-polarised incident light will preferentially photoexcite chromophores with their absorption transition moments orientated perpendicular to the interface. The anisotropy expressions for the two excitation polarisation conditions are different (to each other the bulk solution situation), and these expressions can be extended to the time-resolved regime; for in- and out-of-plane processes, the fluorescence anisotropy is given by:
where ss corresponds to s-polarised excitation and s-polarised emission, sp = s-polarised excitation and p-polarised emission and ps = p-polarised excitation and s-polarised emission.
So if a series of fluorescence decay measurements are performed under appropriate polarisation conditions the timescales of the processes inducing changes in fluorescence anisotropy in- and out-of- the plane of the interface can be monitored.
Apparatus:
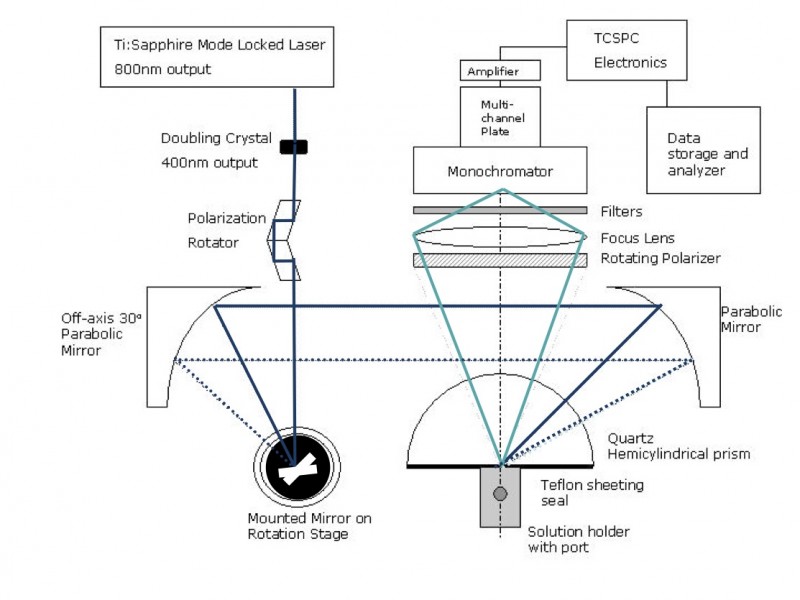
Below is a schematic of our previous time-resolved, polarised evanescent wave experimental setup. Having two parabolic mirrors after the rotating mirror allows the angle of incidence to be varied (and thus the excitation depth) without causing misalignment of the detection system.
We found that this configuration worked reasonably well, but the alignment was a little troublesome. We have therefore recently modified our rig so we can perform variable angle, polarised, time-resolved EWIF measurements.
Relevant papers:
- C.A. Scholes, D.P. Millar, M.L. Gee and T.A. Smith, “Resonance Energy-Transfer Studies of the Conformational Change on the Adsorption of Oligonucleotides to a Silica Interface”, J. Phys. Chem. B (2011) 115 (19), 6329–6339
- A.C. Rapson, A. Hossain, J.D. Wade, E.C. Nice, A.H.A. Clayton, T.A. Smith and M.L. Gee, “Structural Dynamics of a Lytic Peptide Interacting with a Supported Lipid Bilayer”, Biophysical Journal 100, 1353-1361 (2011)
- T.A. Smith, C.A. Scholes and M.L. Gee, “Polarized Time-Resolved Evanescent Wave-Induced Fluorescence Measurements”, in Reviews in Fluorescence, C.D. Geddes and J.R. Lakowicz (Eds), Springer Science, pp 245-270, (2005)
- M.L. Gee, L. Lensun, T.A. Smith and C.A. Scholes, “The Determination of Molecular Conformational Changes of Proteins at an Interface Using Time-Resolved Evanescent Wave-Induced Fluorescence Anisotropy Measurements”, Eur. Biophys. J, (2004), 33(2), 130-139
- M.L. Gee and T.A. Smith, “Shedding Light on the Conformation of Proteins and Other Macromolecules at an Interface”, Aust. J. Chem. (2003), 56, 1005-1012